Differential Impact of Ventricular-Arterial Coupling on Left Ventricular Function in Patients with Acute Myocardial Infarction: A Comparison Between Preserved and Reduced Ejection Fraction
Jeju University College of Medicine Hae Eun Yun
Journal of Cardiovascular Imaging (2025) 33:2
Ventricular-arterial coupling (VAC) plays a critical role in the development and progression of heart failure in patients with coronary artery disease. VAC affects both systolic and diastolic function of the left ventricle, and the underlying mechanisms are known to differ between heart failure with reduced ejection fraction (HFrEF) and preserved ejection fraction (HFpEF). Accordingly, the impact of ventriculo-arterial coupling (VAC) on left ventricular function in patients with coronary artery disease may vary depending on ejection fraction (EF). However, research on this topic remains limited. Therefore, this study aimed to investigate the relationship between VAC and left ventricular function by classifying patients with acute myocardial infarction (AMI) according to their EF.
Standard echocardiographic techniques were used to assess LV volume and systolic and diastolic function. Effective Arterial Elastance (EA) was calculated based on stroke volume derived from the LV outflow tract waveform, while Effective End-Systolic Elastance (ELV) was estimated using the single-beat method.
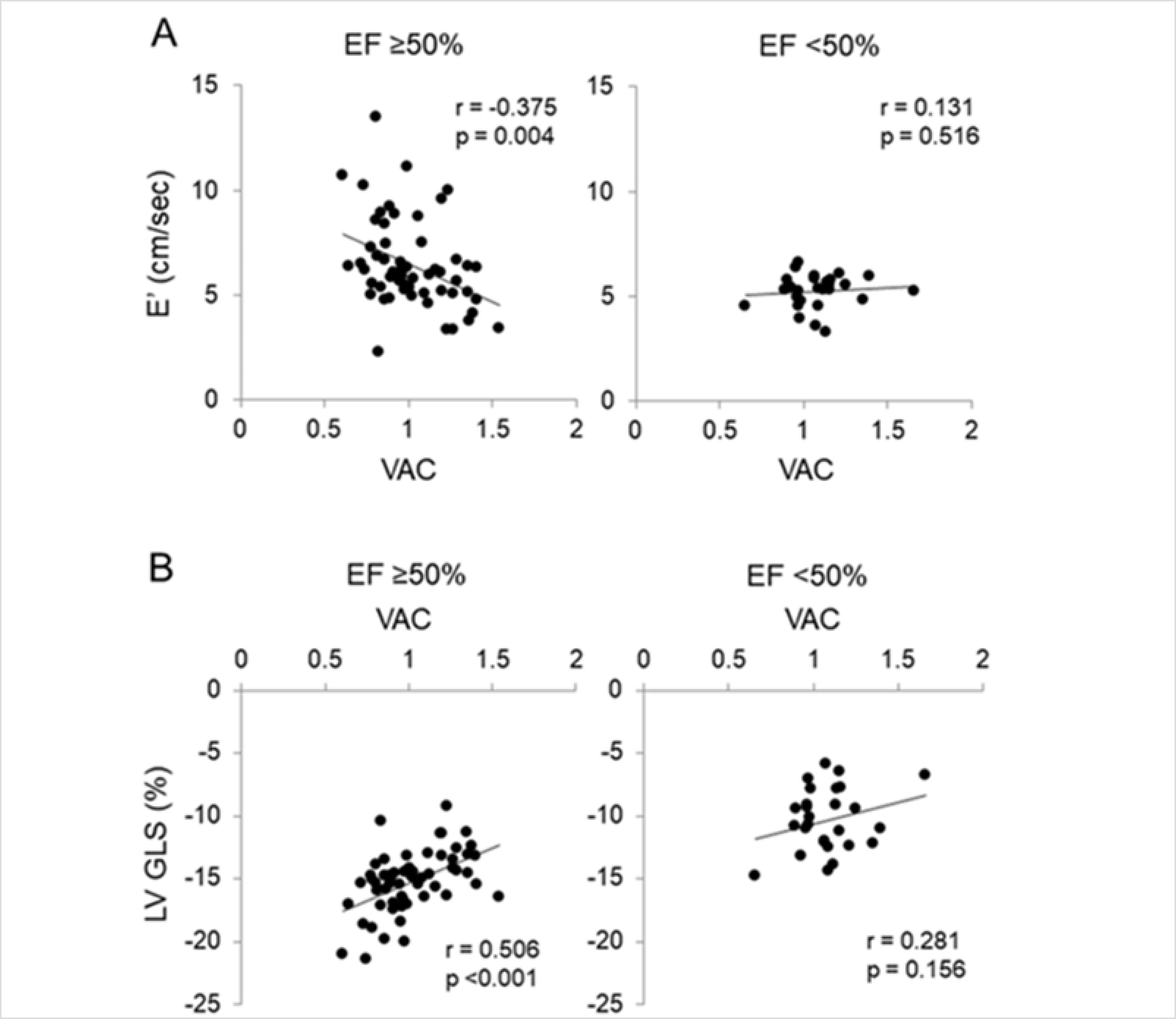
A total of 85 patients with acute myocardial infarction (AMI) were included in the study (mean age 58.5 ± 10.6 years). Based on their ejection fraction (EF), patients were categorized into a reduced EF group (EF < 50%; n=27) and a preserved EF group (EF ≥ 50%; n=58). In the adjusted linear regression analysis, the preserved EF group showed a significant negative correlation between E′ velocity and VAC (β = -0.310; P = 0.008), whereas no such association was observed in the reduced EF group. Additionally, LV global longitudinal strain (GLS) had a significant positive correlation with VAC (β = 0.505; P < 0.001), EA (β = 0.536; P < 0.001), and Zc (β = 0.344; P = 0.018) only in the preserved EF group. These findings suggest that in AMI patients with preserved EF, aortic load and ventricular-arterial mismatch may contribute to the progression toward HFrEF, whereas their influence appears to be limited once EF is already reduced. Therefore, in AMI patients with preserved EF, assessment of aortic load and VAC is important for predicting and potentially preventing progression to HFrEF. In particular, as antihypertensive treatment can reduce arterial stiffness and improve VAC, stricter blood pressure control may be necessary in AMI patients with coexisting hypertension.
In conclusion, these findings confirmed that the relationship between hemodynamic parameters and both diastolic and systolic function of the left ventricle differs depending on EF status. Moreover, the mechanical interaction between the ventricle and arterial system varies according to the level of left ventricular systolic function.